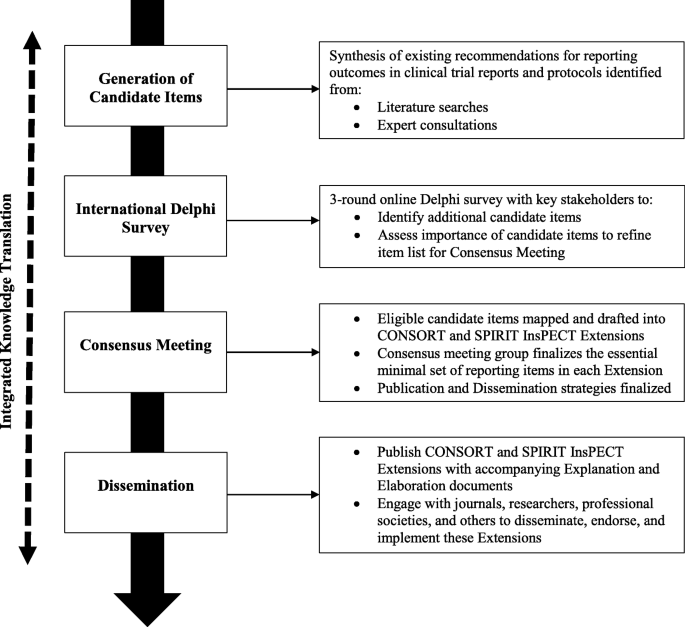
Comparison of serious adverse events posted at ClinicalTrials.gov and published in corresponding journal articles – The Publication Plan for everyone interested in medical writing, the development of medical publications, and publication planning
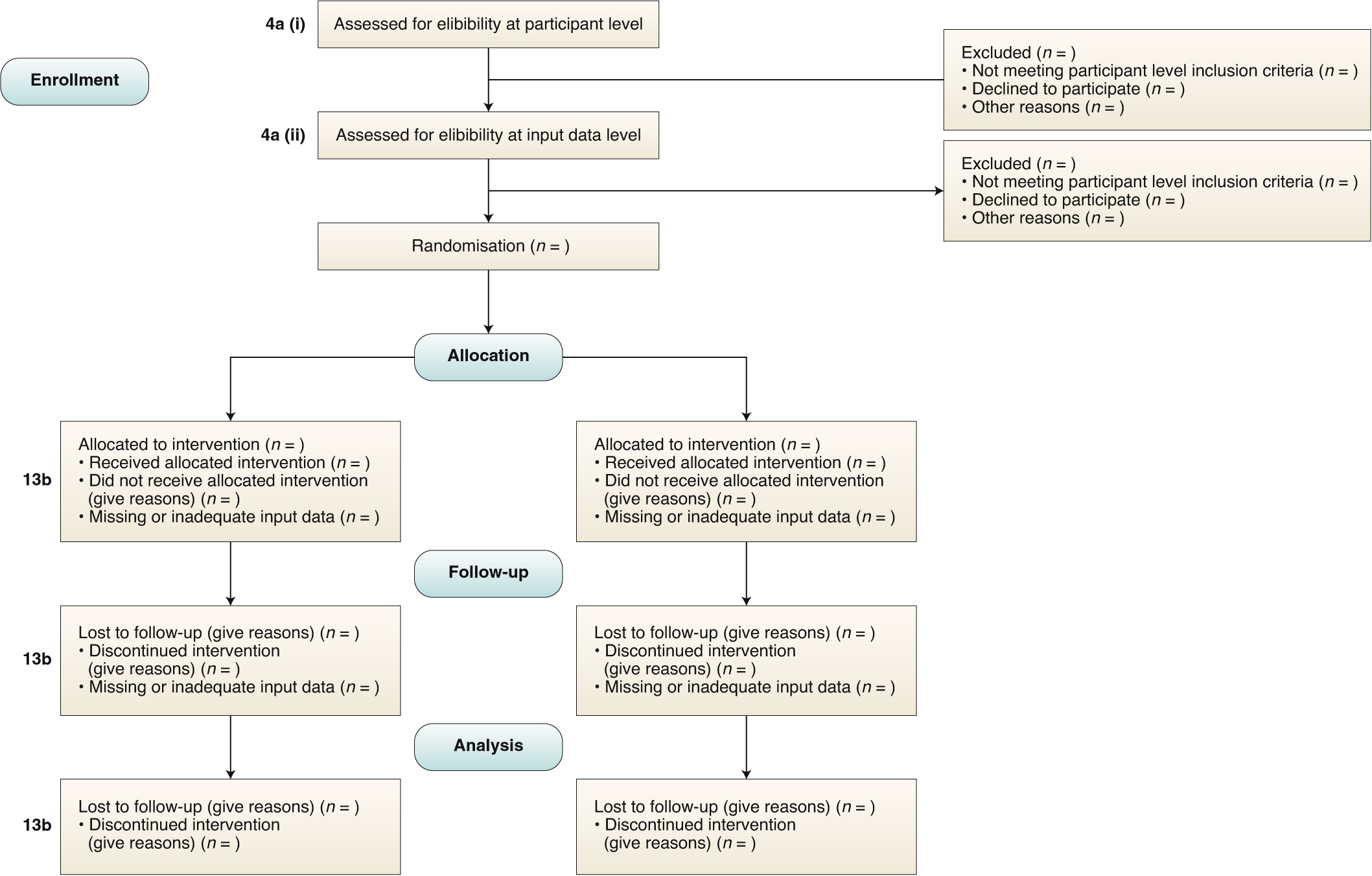
Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension | Nature Medicine

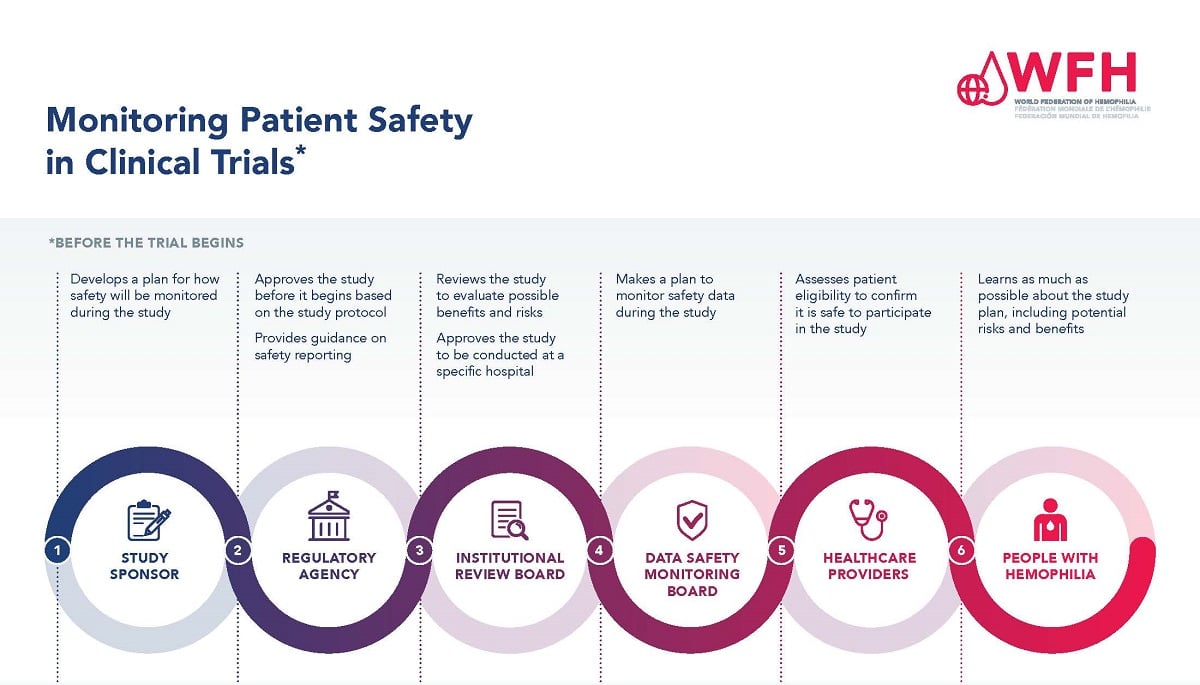
Global Healthcare Brand Improves Safety Reporting in Clinical Trials Leveraging Pharmacovigilance Analytics| Quantzig | Business Wire

1 Nuts and Bolts of Safety Reporting The Role of the CRO Dr. Noa Lowenton Spier Pharma-Clinical S.A.G. - ppt download

Process of reporting serious adverse events (SAE) during a regulatory... | Download Scientific Diagram

Clinical Trials: A Practical Guide to Design, Analysis and Reporting: Wang, Duolao: 9781901346725: Amazon.com: Books

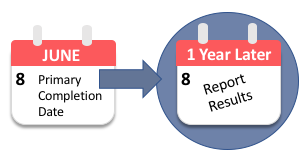
Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet

Randomised Clinical Trials: Design, Practice and Reporting: 9781119524649: Medicine & Health Science Books @ Amazon.com

After years of lax oversight, the NIH is starting to contact institutions about unreported clinical trial results - STAT