Buy Becoming a Successful Clinical Trial Investigator: A Step by Step Guide for Developing a Clinical Trial Site Book Online at Low Prices in India | Becoming a Successful Clinical Trial Investigator:
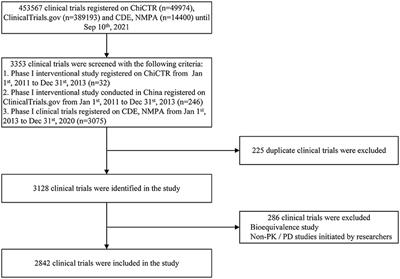
Frontiers | Trends of Phase I Clinical Trials of New Drugs in Mainland China Over the Past 10 Years (2011–2020)

Challenges of phase 1 clinical trials evaluating immune checkpoint-targeted antibodies - Annals of Oncology

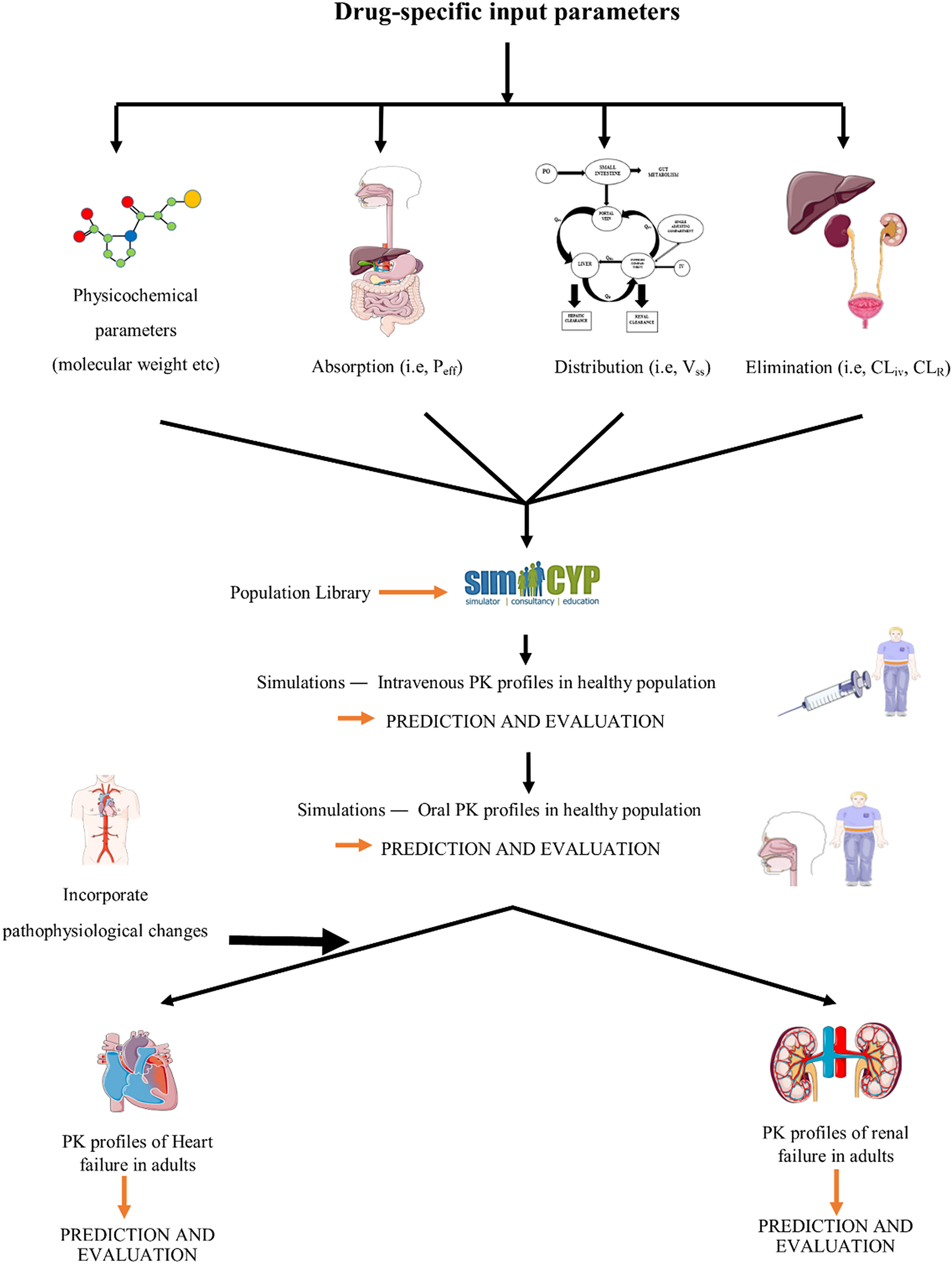
Current status and future perspective on preclinical pharmacokinetic and pharmacodynamic (PK/PD) analysis: Survey in Japan pharmaceutical manufacturers association (JPMA) - ScienceDirect

Optimal biological dose: a systematic review in cancer phase I clinical trials | BMC Cancer | Full Text

Metformin Clinical DDI Study Design That Enables an Efficacy- and Safety-Based Dose Adjustment Decision | Pharmaron
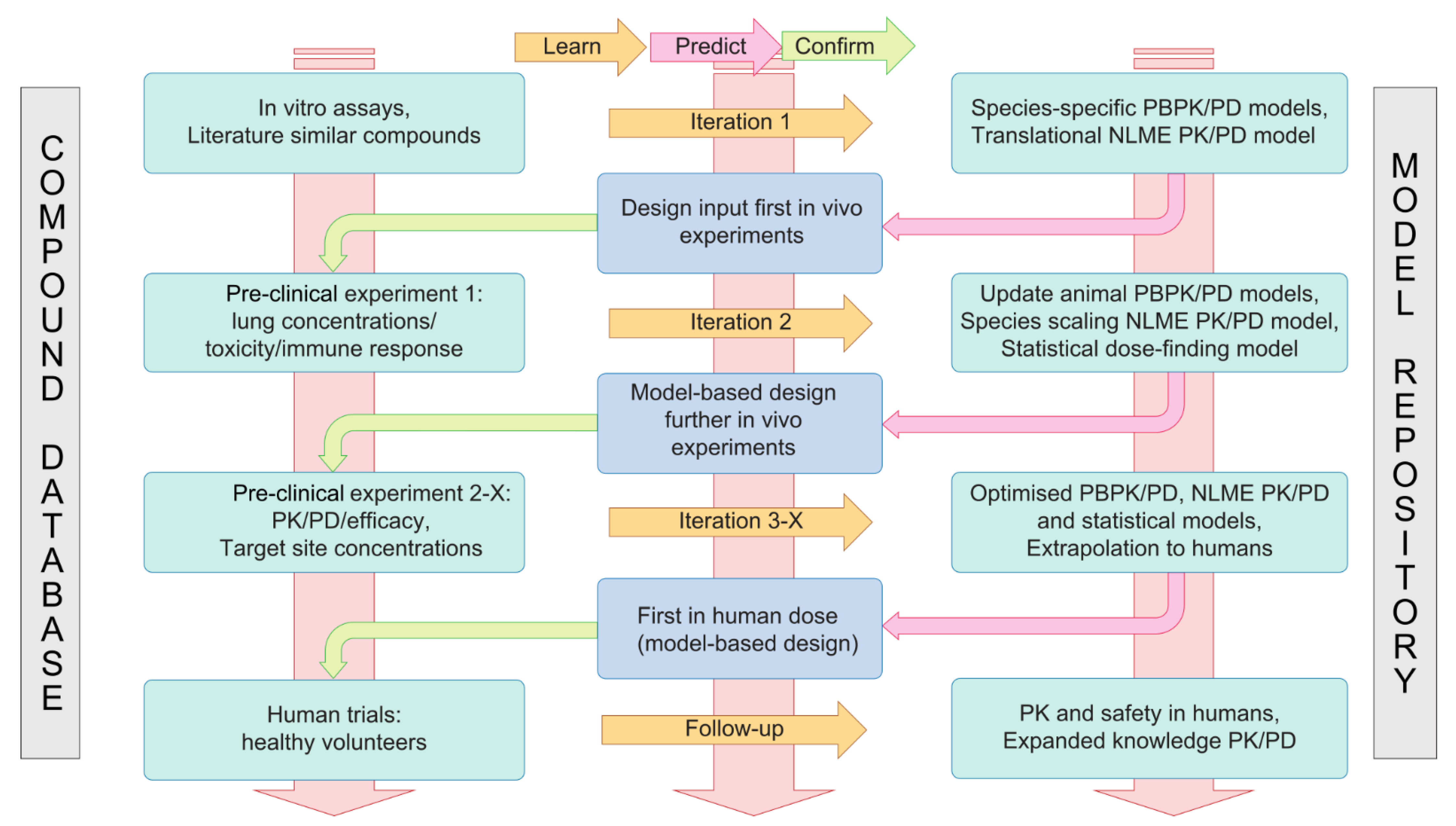
Pharmaceutics | Free Full-Text | The Use of Translational Modelling and Simulation to Develop Immunomodulatory Therapy as an Adjunct to Antibiotic Treatment in the Context of Pneumonia

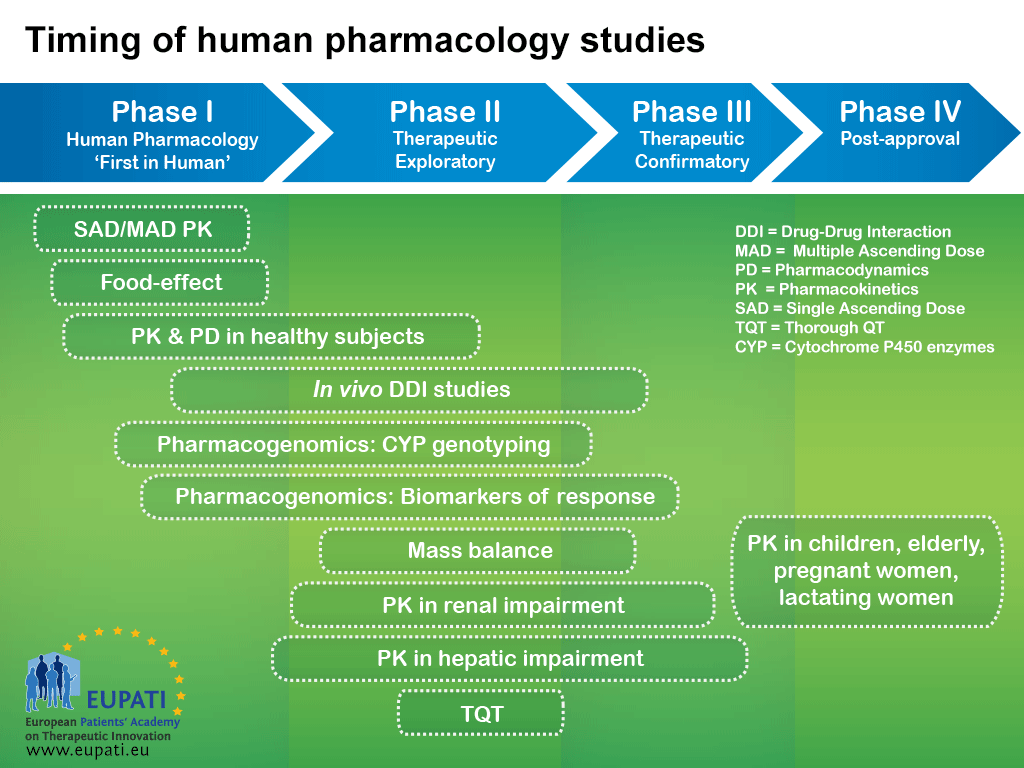
Pharmacokinetic and Statistical Considerations in First-in-Human Clinical Trials | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services

Pharmacokinetic and Statistical Considerations in First-in-Human Clinical Trials | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services

10/20/2015 - Session II: Clinical Trial PK/PD Translated To Population Drug Use and Exposure - YouTube



![PDF] Phase 0 Clinical Trial Strategies for the Neurosurgical Oncologist | Semantic Scholar PDF] Phase 0 Clinical Trial Strategies for the Neurosurgical Oncologist | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ba643ff4c798a7864e6a78f009413a89e764a2c0/6-Figure2-1.png)