Targeting non-canonical pathways as a strategy to modulate the sodium iodide symporter - ScienceDirect
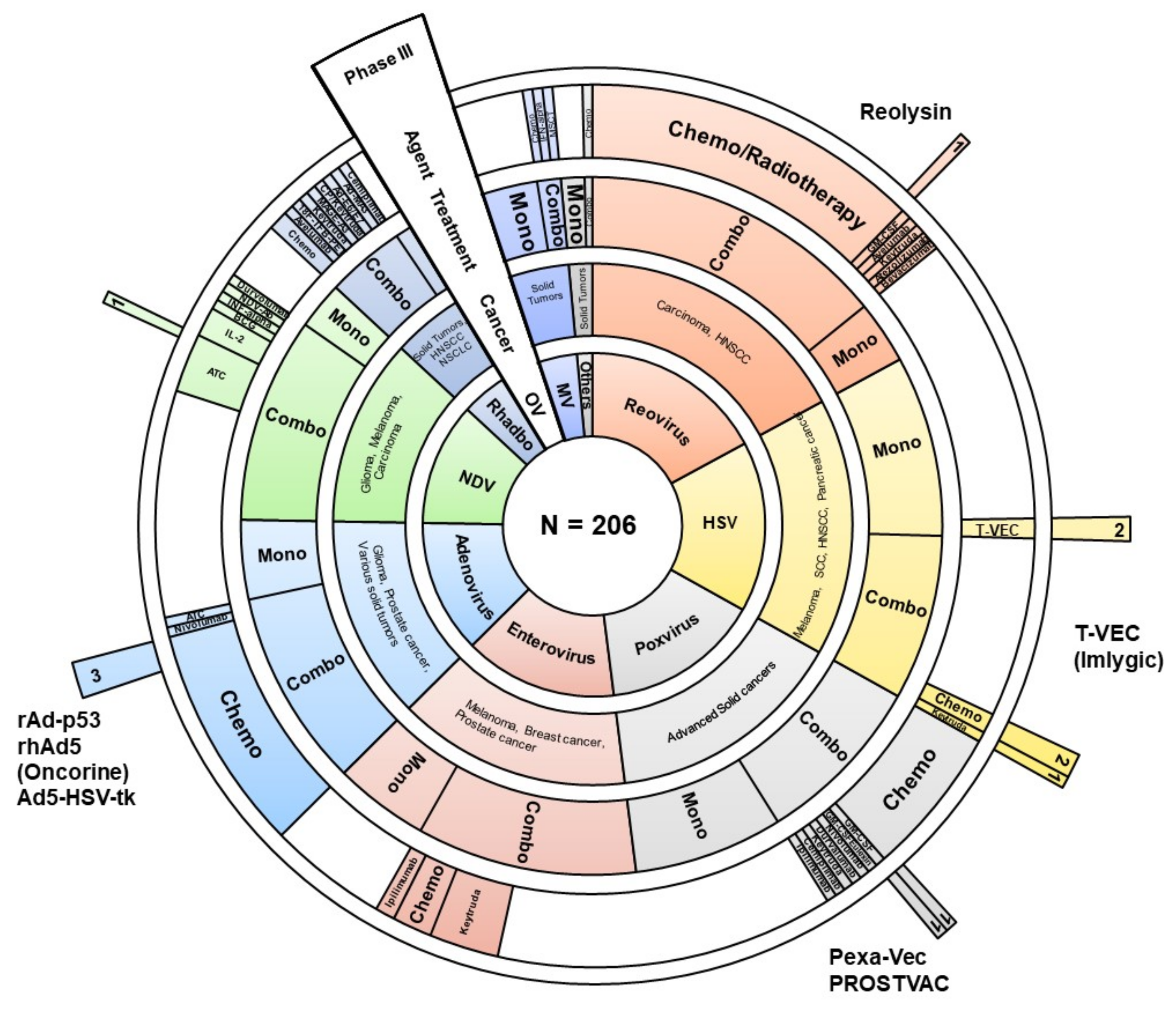
Viruses | Free Full-Text | Combinatorial Approaches for Cancer Treatment Using Oncolytic Viruses: Projecting the Perspectives through Clinical Trials Outcomes

Interventional vs. Non-interventional Study Classification in the EU: Considerations on the Impact of Direct-to-Patient Contacts