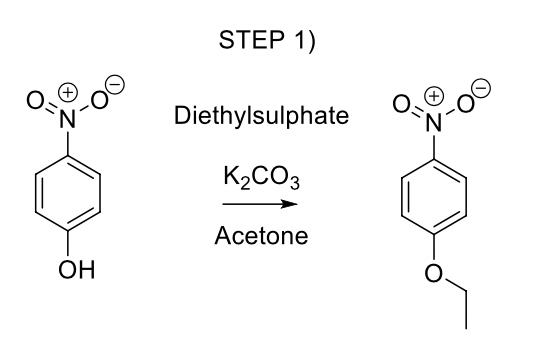
When acetone reacts with dilute aqueous solution of Ba OH 2, the product obtained is calledA. diacetone alcoholB. PhoroneC. crotonaldehydeD. Acetophenone
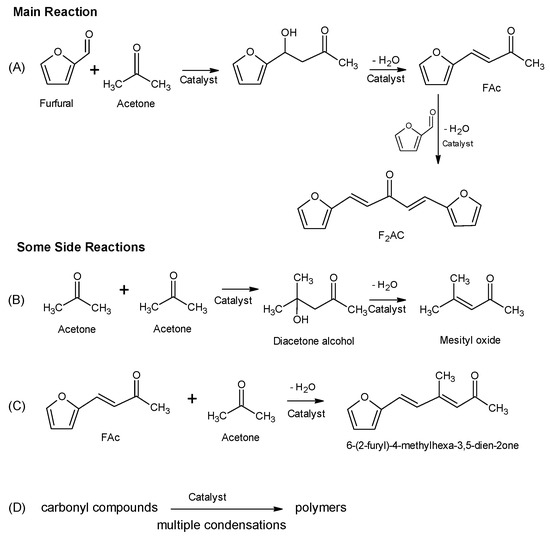
Catalysts | Free Full-Text | Aldol Condensation of Furfural with Acetone Over Mg/Al Mixed Oxides. Influence of Water and Synthesis Method

Competitive Adsorption of Methanol–Acetone on Surface Functionalization (−COOH, −OH, −NH2, and −SO3H): Grand Canonical Monte Carlo and Density Functional Theory Simulations | ACS Applied Materials & Interfaces

Potential energy profiles for the OH + acetone reaction system using... | Download Scientific Diagram

Reaction of Hydroxyl Radical with Acetone. 2. Products and Reaction Mechanism | The Journal of Physical Chemistry A
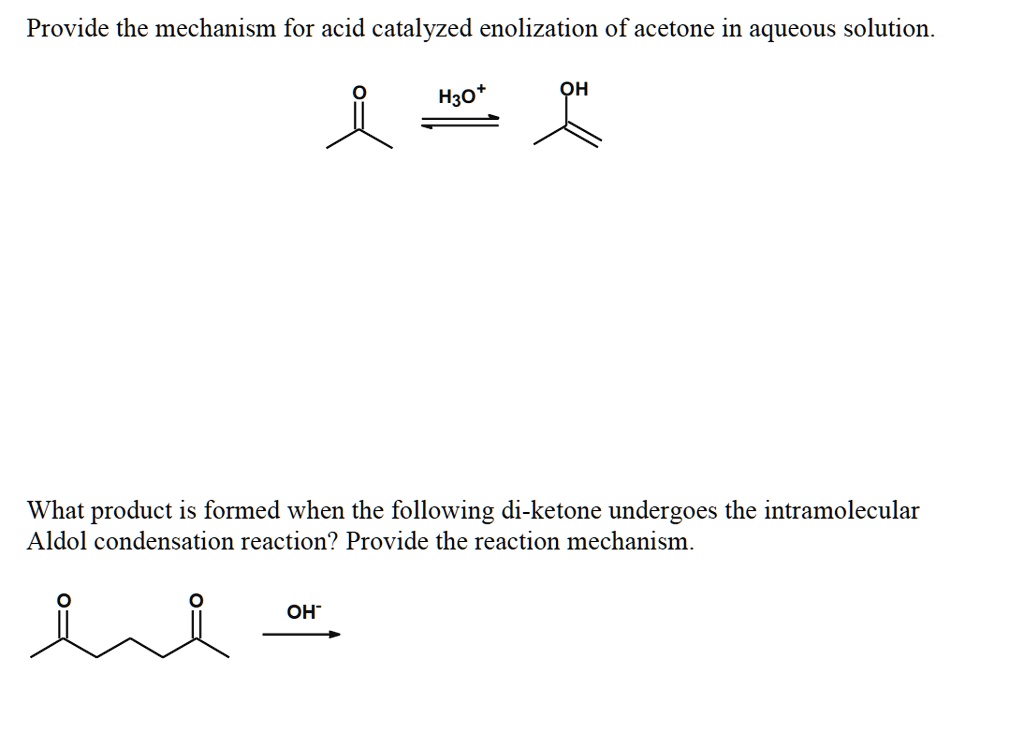
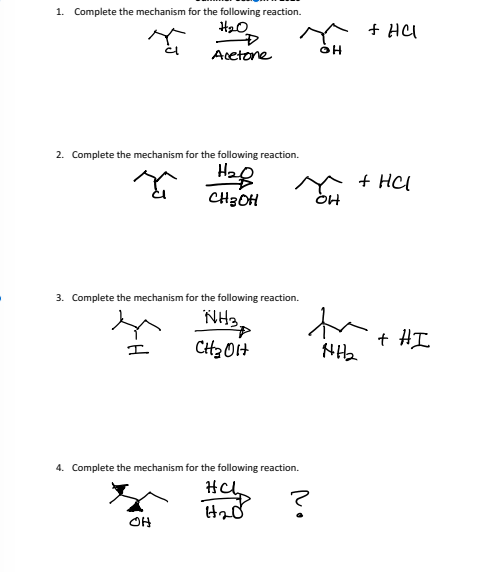
SOLVED: Provide the mechanism for acid catalyzed enolization of acetone in aqueous solution. H3O OH What product is formed when the following di-ketone undergoes the intramolecular Aldol condensation reaction? Provide the reaction
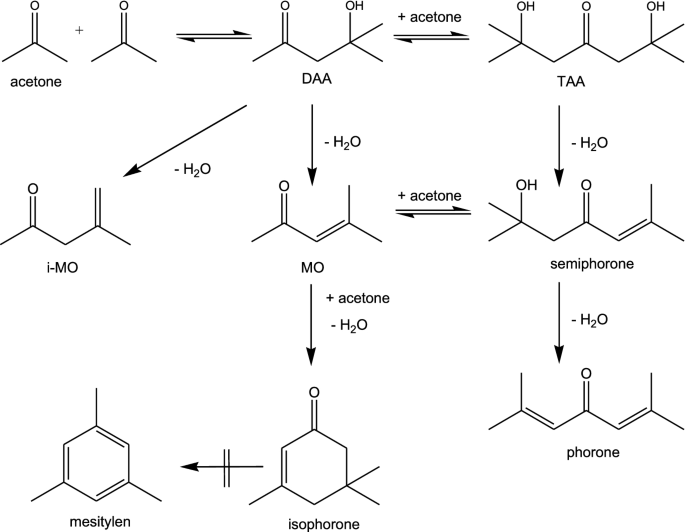
Upgrading bio-based acetone to diacetone alcohol by aldol reaction using Amberlyst A26-OH as catalyst | SpringerLink

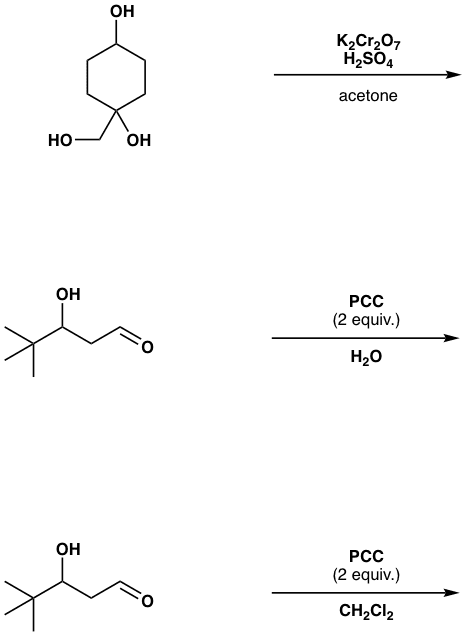
Devise a synthesis of the below compound using acetone (CH3)2C=O is the only source of carbon atoms. You may use any needed organic or inorganic reagents. | Homework.Study.com

The product formed when acetone is heat with `Ba(OH)_(2)` is : - Sarthaks eConnect | Largest Online Education Community